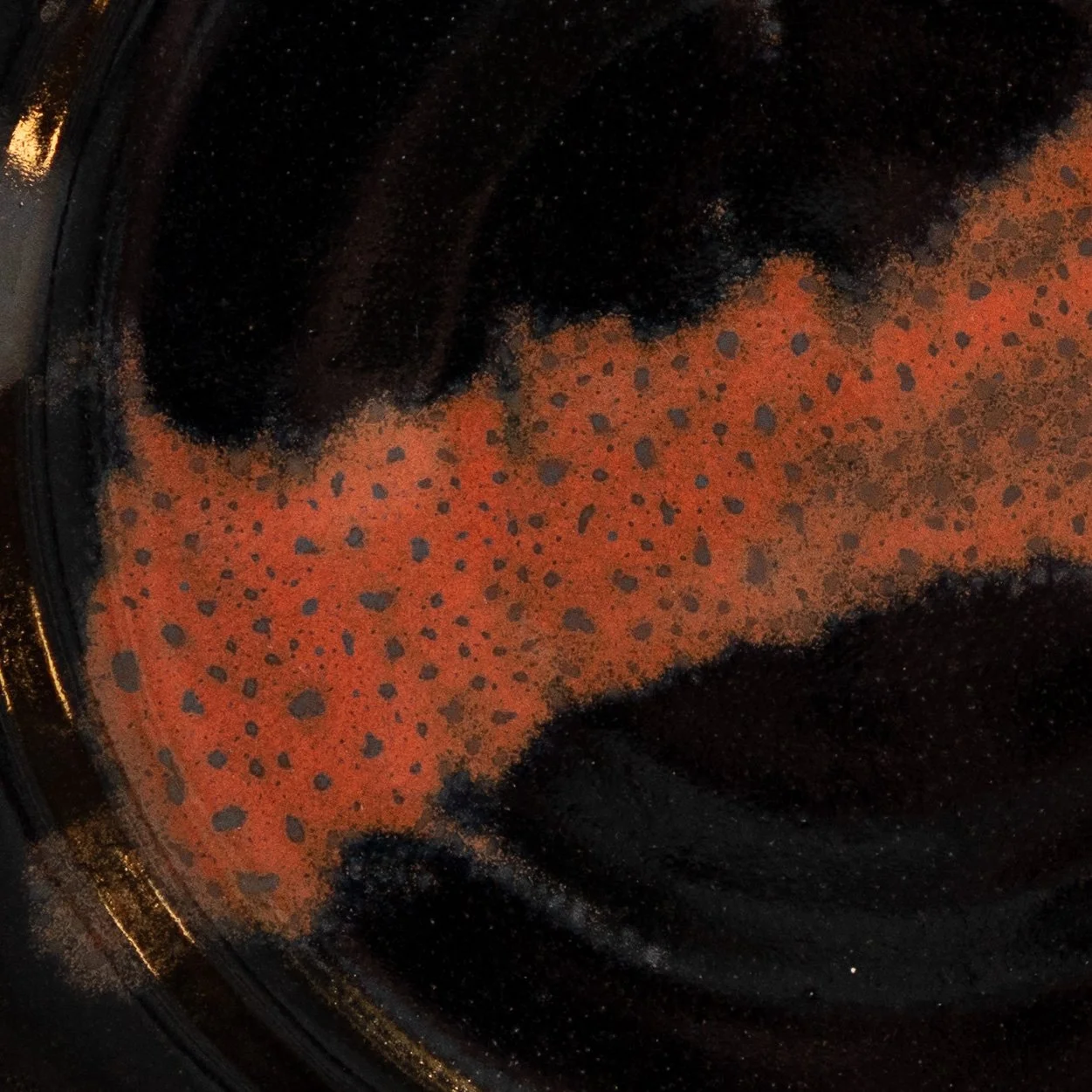
Iron Saturate Glazes
During the next few days I'll be releasing a series showing how to create a glaze using Glazy and volumetric blending.
The first step is familiarizing oneself with the glaze type. For this demonstration I'm interested in creating a Cone 10 Iron-Saturate Red microcrystalline glaze also known as "Kaki", "Tomato Red", and "Persimmon". Some historically examples are Chinese Song Dynasty Ding Persimmon-glazed wares as well as many of Shoji Hamada's works.
For each search, Glazy shows Recipe Cards with photos as well as a Stull Chart. The Stull Chart has five major regions: Unfused, Matte, Semi-Matte, Bright/Gloss, and Under-fired. There is another area, Crazed, that overlaps the other regions. In the next step, I will create a Biaxial Test using the Stull chart as a guide.
From the analyses of Iron-Saturate glazes in Glazy, it is not clear what the ideal amount of Silica and Alumina (and the Si:Al ratio) should be. So our first step will be to re-create the Si:Al Stull Chart with a prototype Iron-Saturate glaze. In an Si:Al grid we can only adjust the amounts of Silica and Alumina, so we must set in stone the other characteristics of the glaze. Looking at the analyses of recipes in Glazy, it is apparent that we will need a good deal of Iron as well as Phosphorus. For our fluxes, apparently some MgO is required. As an educated guess, or initial prototype Iron Saturate glaze will have variable Silica and Alumina, while the following are set for all glazes: KNaO 0.2, CaO 0.6, MgO 0.2, Fe2O3 0.22, P2O5 0.12.
We will use volumetric blending to magically create 25 glaze tests from only 4 batches of glaze. The four corner glazes composed using the Glazy Recipe Calculator. The columns originate from the Origin so that each column represents a specific Si:Al ratio. It is decided to put the "Left" column in the Stull Matte Region, while the "Right" column is pushed close to the Under-Fired Region.
Batches of 500 grams are created for each corner glaze, and the test glazes are mixed using volumetric blending with samples of 20mg. The tests are ready to be fired!
Note: The "educated guess" for our initial prototype glaze is informed in large part by the amazing work of Carol Marians. Carol has posted 8 years of glaze research on her website at:
I made a mistake on the Si:Al ratios for each biaxial column. The ratios should be approximately 4.3, 5.4, 7, 9.3, and 12.8
Here are the results of the Iron-Saturate Biaxial. While the reduction firing with uncontrolled cooling results in brown, metallic surfaces, the oxidation firing with a controlled cool and hold at 1700°F (925°C) gives us more interesting results. The biaxial reveals that an Si:Al ratio of around 9-9.5 (the fourth column) promotes redder glazes. In particular, tile D4 (4th row, 4th column) seems promising.
The oxidation firing schedule is adopted from Carol Marians, but simplified to:
150°F/hr to 250°F (65°C/hr to 120°C)
400°F/hr to 2050°F (200°C/hr to 1120°C)
120°F/hr to 2250°F (50°C/hr to 1230°C)
60°F/hr to 2290°F (16°C/hr to 1250°C)
40°F/hr to 2310°F (4°C/hr to 1265°C)
Hold of 10 minutes at 2310°F (1265°C)
400°F to 1700°F (925°C) - Down-fire
Hold 2 hours at 1700°F (925°C)
See http://carol.knighten.org for many more examples of firing schedules for iron red glazes.
Step 2: Refine Silica:Alumina Ratio
From the Iron-Saturate Biaxial we choose tile D4 to work with. We can now "zoom in" and refine the Si:Al ratio for this tile. At Si:Al 8-8.5 the glaze seems more evenly covered in crystals. As the Si:Al ratio is increased the coverage becomes more splotchy.
Step 3: Refine Flux Ratio
It seems the Si:Al ratio for biaxial tile D4 was already pretty good. Now we can move on to testing factors other than Si:Al in the prototype glaze. In this test, we increase the level of R2O (KNaO, or K2O & Na2O) while decreasing the amount of Calcium. I was surprised by the result for 0.3 KNaO, perhaps there would have been a better result if both Calcium and Magnesium were decreased? Or decrease Si & Al? Anyway, based on this test I'll just stay at R2O:RO 0.2:0.8
Step 4: Refine Alkaline Fluxes
Test of Iron-Saturate Biaxial tile D4 replacing Mahavir Potash Feldspar with Minspar 200 Soda Feldspar. Not a 1-to-1 percentage replacement, but maintaining the same UMF (except K2O and Na2O).
Step 5: Refine Alkaline Earth Fluxes
Now that we've established an R2O:RO ratio of 0.2 using Potash Feldspar, we can test the best proportion of Calcia to Magnesia. With our R2O set at 0.2, 0.8 remains for the RO (including Calcia and Magnesia) portion of our UMF fluxes. The educated guess of 0.6 CaO and 0.2 MgO in our original Iron Saturate biaxial turns out to be a good choice. More than 0.2 MgO also gives some interesting glazes with a more metallic surface.
Step 6: Test different sources of MgO
Same UMF, different sources of Magnesia.
Step 7: Iron & Bone Ash Biaxial
Glazes are often split into two parts: A base glaze and additives like colorants and opacifiers. It's like ordering a pizza (base) with toppings (additives). We've already tested many aspects of the Iron-Saturate base glaze including Silica:Alumina, R2O:RO, and Calcia:Magnesia. Now we can move on to the additives: Iron and Bone Ash. (The decision between what is part of the base recipe vs. an additive is somewhat arbitrary. Just as with a pizza that's made it to your stomach, it all eventually ends up mixed together.) Additives are added in addition to the base glaze recipe. So in this test, I did not alter the base glaze at all, the only difference is increasing Red Iron Oxide and Bone Ash. I was surprised to see that additional bone ash didn’t alter the glaze a lot more.
Step 8: Bone Ash Line Blend
A line blend of biaxial test D4 blended with the same recipe without Bone Ash (but maintaining the same fluxes to account for the missing CaO from the Bone Ash). Without P2O5, our glaze is a nice tenmoku. At 0.03 P2O5 very faint traces of crystallization appear. At 0.06 P2O5 crystallization is much more evident, and somewhere between 0.06 and 0.09 P2O5 there is a dramatic transformation.
Step 9: Source of Iron Oxide
Testing different sources of iron using tile D4 from the Iron-Saturate Biaxial. I'm not sure what's going on with Yellow Iron Oxide. It would be interesting to see other sources of iron, especially iron phosphate.
Step 10: Influence of Titania
Adding Titanium Dioxide in 1% increments to tile D4 from the Iron-Saturate Biaxial. Titanium Dioxide is often present in analyses of Song Dynasty Russet/Persimmon glazes as well as Japanese Kaki glazes, but usually in amounts of less than 1%.
Step 11: Make Stuff
A variation of the glaze from the original biaxial: https://glazy.org/recipes/25001